CLINICAL TOOLS
Welcome to the Clinical Tools section of the Eczema Hub! This section is designed for clinicians to find the most commonly used, established, validated, and reliable clinical tools for patients presenting with atopic dermatitis. These tools are used to support the measurement, diagnosis, and management of atopic dermatitis. It is important to understand that no single tool is sufficient to characterize the severity of atopic dermatitis and it is recommended that a combination of these tools be used to achieve a balanced understanding and representation.
Eczema Area and Severity Index (EASI)
The Eczema Area and Severity Index (EASI) is a validated and consistent scale used to assess the severity and extent of atopic dermatitis (AD). The EASI assesses the severity of four characteristics of AD including erythema, infiltration and/or papulation, excoriations, and lichenification. Scores on this tool range from 0 to 72 points, where higher scores indicate greater severity.
For more comprehensive information on this tool from a Canadian source, click here and explore appendix four (pages 118-120). Despite not being a recent publication, click here for the best and most accurate access to this tool with figures, examples on how to use the tool, and information on the original publication.
Eczema Area and Severity Index (EASI) (Tofte et al., 1998, as cited in Honari, 2017).
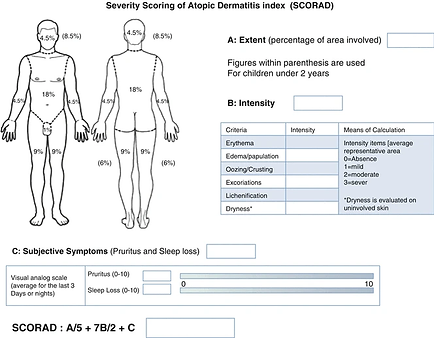
Severity Scoring of Atopic Dermatitis (SCORAD)
Severity Scroing of Atopic Dermatitis (SCORAD), (Severity scoring of atopic dermatitis: the SCORAD index. consensus report of the european task force on atopic dermatitis, 1993, as cited in Honari, 2017).
The Severity Scoring of Atopic Dermatitis (SCORAD) is a valid and reliable tool used to evaluate the extent and severity of AD by looking at three main components: the affected body, severity of clinical signs, and symptoms. The severity of six specific symptoms including redness, swelling, oozing and/or crusting, excoriation, skin thickening and/or lichenification, and dryness are assessed using a four-point scale. The SCORAD is later calculated based on these components. The maximum score achievable is 103 with higher scores indicating a more severe condition.
For more comprehensive information on this tool from a Canadian source, click here and explore appendix four (pages 117-120). Despite not being a recent publication, click here for the best and most accurate access to this tool with figures, information on how to use this tool, and information on the original publication.
Patient-Oriented Eczema Measure (POEM)
The Pateint-Oriented Eczema Measurement (POEM) tool is a seven-item questionnaire used to assess disease symptoms in children and adults. Based on the frequency of occurrence during the past week, seven items including dryness, itching, flaking, cracking, sleep loss, bleeding, and weeping are assessed on a five-point scale. The maximum POEM score is 28. A higher POEM score indicates a poorer quality of life and a greater severity in condition.
For more comprehensive information on this tool from a Canadian source, click here and explore appendix four (pages 119-124). Despite not being a recent publication, click here for the best and most accurate access to this tool with information on how to use the tool, the questions that are asked, and information on the original publication.
Patient-Oriented Eczema Measure (POEM) (Charman et al., 2004, as cited in Honari, 2017).
Investigator's Global Assessment (IGA)
Investigator's Global Assessment (IGA), (International Eczema Council 2024, Copyright ©2017 Eli Lilly and Company – Used with the permission of Eli Lilly and Company under a Creative Commons Attribution-NoDerivatives 4.0 International License - https://creativecommons.org/licenses/by-nd/4.0/).
The Investigator's Global Assessment (IGA) is a validated five-point assessment scale that provides a global clinical assessment of AD. On this scale, a score of “0” indicates clear, and a score of “4” indicates severe AD.
It is important to note that, although this tool in commonly used in clinical trials, a more recent source from this year, found by clicking here, states this tool is feasible in clinical practice.
For more comprehensive information on this tool from a Canadian source, please click here and explore appendix four (pages 118-120). For access to this tool please click here.
Pruritus Numerical Rating Scale (PRNS)
The Pruritus Numerical Rating Scale (PRNS) is a valid and reliable tool used by patients to report the intensity of their itch during a daily recall period. Patients are asked to rate their average and maximum intensity of itch experienced during the past 24 hours based on a scale of 0 (no itch) to 10 (worst itch imaginable).
For more comprehensive information on this tool from a Canadian source, please click here and explore appendix four (pages 118-121). To access the validated version of this tool please click here.
Dermatology Life Quality Index &
Children's Dermatology Life Quality Index
(Copyright ©1992 Dermatology Life Quality Index. A., Y, Finlay, G., K, Khan).
(Copyright ©1993 Children's Dermatology Life Quality Index. M., S. Lewis-Jones, A., Y. Finlay. June 1993 Illustrations ©Media Resources Centre, UWCM. Dec 1996).
The Dermatology Life Quality Index and the Children's Dermatology Life Quality Index are two widely used dermatology-specific quality-of-life instruments. They both employ a 10-item questionnaire that assesses aspects of AD that can affect quality of life.
For more comprehensive information on these tools from a Canadian source, please click here and explore appendix four (pages 118-123). For information and direct access to these tools, please click here and navigate the website.
References
Cardiff University. (2024). Quality of life questionnaires. Cardiff University School of Medicine. https://www.cardiff.ac.uk/medicine/resources/quality-of-life-questionnaires
Canadian Agency for Drugs and Technologies in Health. (2020). CADTH common Drug Review: Clinical Review Report: Dupilumab (Dupixent): (Sanofi Genzyme, a division of sanofi-aventis Canada Inc.): Indication: Indicated for the treatment of patients aged 12 years and older with moderate-to-severe atopic dermatitis whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable (Appendix 4). Ottawa (ON). https://www.ncbi.nlm.nih.gov/books/NBK565237/
Charman CR, Venn AJ, Williams HC. The patient-oriented eczema measure: development and initial validation of a new tool for measuring atopic eczema severity from the patients’ perspective. Arch Dermatol. 2004; 140(12):1513–9.
Finlay, A. Y., & Khan, G. K. (1994). Dermatology Life Quality Index (DLQI)--a simple practical measure for routine clinical use. Clinical and experimental dermatology, 19(3), 210–216. https://doi.org/10.1111/j.1365-2230.1994.tb01167.x
Honari, G. (2017). Clinical Scoring of Atopic Dermatitis. In: Humbert, P., Fanian, F., Maibach, H., Agache, P. (eds) Agache's Measuring the Skin. Springer, Cham. https://doi.org/10.1007/978-3-319-32383-1_94
International Eczema Council. (2024). Investigator Global Assessment Scale. Eczema Council. https://www.eczemacouncil.org/index.php?option=com_content&view=article&id=49:investigator-global-assessment-scale&catid=20:site-content&Itemid=136.
Lewis-Jones, M. S., & Finlay, A. Y. (1995). The Children's Dermatology Life Quality Index (CDLQI): initial validation and practical use. The British journal of dermatology, 132(6), 942–949. https://doi.org/10.1111/j.1365-2133.1995.tb16953.x
Narla, S., & Silverberg, J. I. (2024). Which Clinical Measurement Tools for Atopic Dermatitis Severity Make the Most Sense in Clinical Practice?. Dermatitis : contact, atopic, occupational, drug, 35(S1), S13–S23. https://doi.org/10.1089/derm.2022.0087
Phan, N. Q., Blome, C., Fritz, F., Gerss, J., Reich, A., Ebata, T., Augustin, M., Szepietowski, J. C., & Ständer, S. (2012). Assessment of pruritus intensity: prospective study on validity and reliability of the visual analogue scale, numerical rating scale and verbal rating scale in 471 patients with chronic pruritus. Acta dermato-venereologica, 92(5), 502–507. https://doi.org/10.2340/00015555-1246
Severity scoring of atopic dermatitis: the SCORAD index. consensus report of the european task force on atopic dermatitis. Dermatology. 1993;186(1):23–31.
Storck, M., Sandmann, S., Bruland, P., Pereira, M. P., Steinke, S., Riepe, C., Soto-Rey, I., Garcovich, S., Augustin, M., Blome, C., Bobko, S., Legat, F. J., Potekaev, N., Lvov, A., Misery, L., Weger, W., Reich, A., Şavk, E., Streit, M., Serra-Baldrich, E., … Zeidler, C. (2021). Pruritus Intensity Scales across Europe: a prospective validation study. Journal of the European Academy of Dermatology and Venereology : JEADV, 35(5), 1176–1185. https://doi.org/10.1111/jdv.17111
Tofte, S.J., Graeber, M., Cherill, R.J., Omoto, M., Thurston, M., & Hanifin, J. (1998). Eczema area and severity index (EASI): A new tool to evaluate atopic dermatitis. Journal of The European Academy of Dermatology and Venereology, 11.
Verweyen, E., Ständer, S., Kreitz, K., Höben, I., Osada, N., Gernart, M., Riepe, C., Pereira, M., Blome, C., Augustin, M., & Zeidler, C. (2019). Validation of a Comprehensive Set of Pruritus Assessment Instruments: The Chronic Pruritus Tools Questionnaire PRURITOOLS. Acta dermato-venereologica, 99(7), 657–663. https://doi.org/10.2340/00015555-3158